Trusted by 50+ Pharmaceutical Companies





At IntraHub Africa, our mission is to deliver scalable, compliant, and user-centric solutions that simplify drug safety reporting, empower stakeholders, and protect patients. We are committed to bridging the gap between pharmaceutical companies, regulatory bodies, and healthcare professionals through advanced technology and expert services.
To be Africa's most trusted partner in pharmacovigilance, driving safer healthcare through innovation and collaboration.
To deliver scalable, compliant, and user-centric solutions that simplify drug safety reporting, empower stakeholders, and protect patients.
IntraHub Africa was founded to address a critical gap in African pharmacovigilance. The right expertise existed, but the tools, training, and systems to put it to work were scattered, imported, or missing entirely.

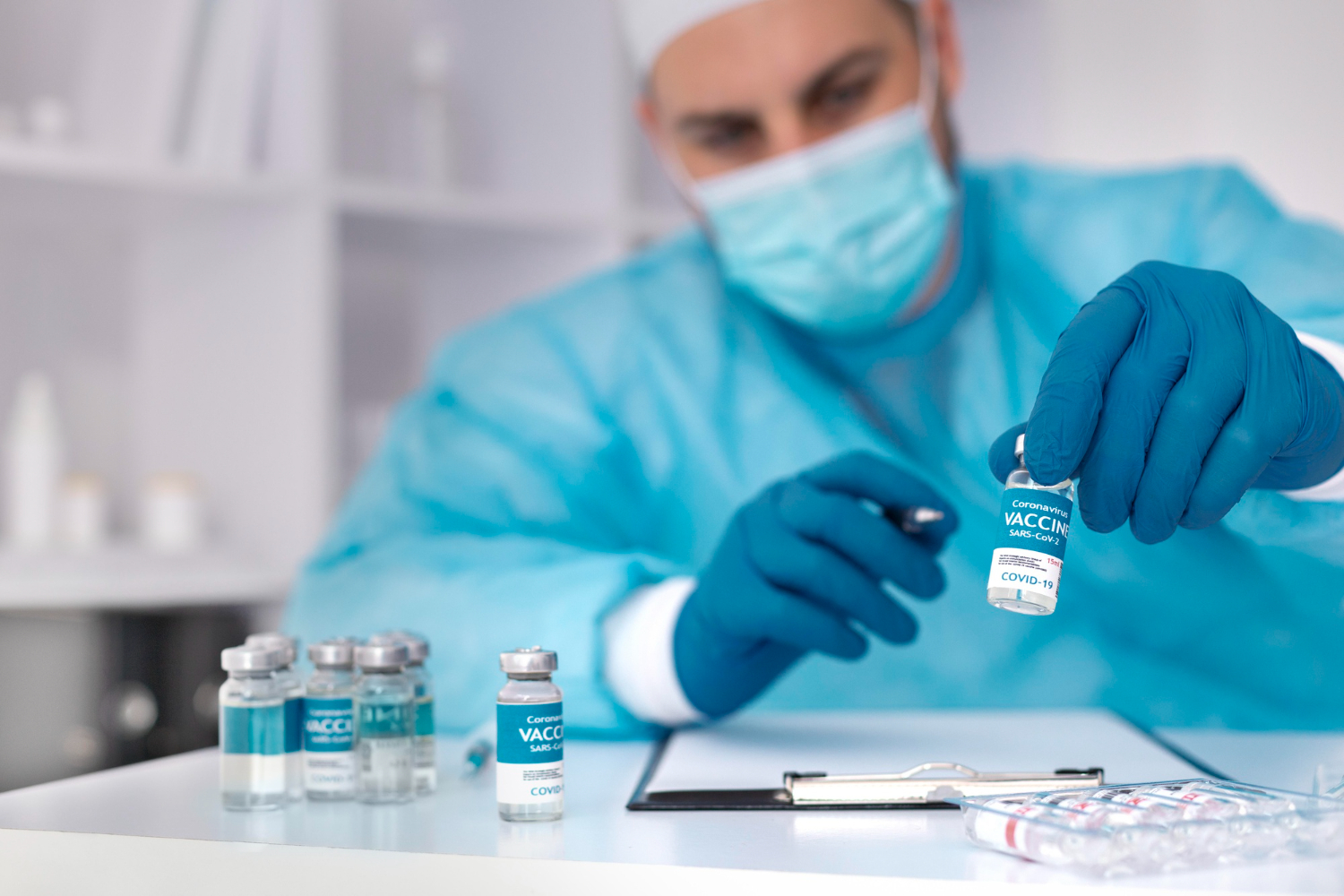
Pharmaceutical companies struggled with paper-based adverse event reporting. Regulators waited on submissions that came late or incomplete. Healthcare workers wanted to report ADRs but had no simple, accessible way to do it.
We set out to fix all of it in one place — combining pharmacovigilance consulting, regulatory affairs support, accredited training, and technology platforms designed from the ground up for African realities.
Today, IntraHub Africa serves pharmaceutical companies, marketing authorisation holders, CROs, and healthcare institutions across Nigeria and the wider African continent.
We combine technological innovation with deep local expertise and a client-centric philosophy to deliver pharmacovigilance solutions that truly work across Africa.
We leverage the latest technology, including AI-driven analytics and real-time monitoring systems, to enhance drug safety and streamline regulatory compliance.
Our solutions are designed with our clients in mind. We work closely with pharmaceutical companies, regulatory bodies, and healthcare organisations to understand their unique needs.
We align with global regulatory frameworks such as ICH and WHO guidelines while maintaining a deep understanding of the local regulatory environment across Africa.
Through our accredited e-learning programs and interactive workshops, we are committed to elevating the pharmacovigilance expertise of healthcare professionals across Africa.
Our values are not aspirations on a wall — they are the operating principles behind every project, every platform, and every partnership we enter.
Value 01
Every decision we make is measured against one question: does this make medicines safer for the people who take them?
Value 02
We follow the evidence, cite the guidelines, and never cut corners on quality. Our work is grounded in GVP, ICH, and local regulatory science.
Value 03
Technology only counts if it works in the field. We build for real conditions, including intermittent connectivity, mobile-first users, and resource-constrained settings.
Value 04
Our clients are partners, not accounts. We succeed when your pharmacovigilance system is compliant, your team is capable, and your patients are safer.
Value 05
We are transparent in our processes, honest about what we can deliver, and accountable for the results.
Watch how our pharmacovigilance technology and regulatory expertise are helping pharmaceutical companies and healthcare institutions build safer, more compliant operations across Africa.
Call Us!
E-mail Us!
Our leadership team brings together deep pharmacovigilance expertise, regulatory affairs knowledge, and technology innovation — all focused on advancing drug safety across Africa.
[Title — e.g. Head of Regulatory Affairs]
[Title — e.g. Chief Technology Officer]

Trusted across Nigeria & West Africa
IntraHub Africa is a pharmacovigilance and regulatory affairs company. We provide PV system setup, ICSR management, regulatory strategy, accredited training, and purpose-built technology platforms — all designed specifically for African pharmaceutical markets.
We are headquartered in Nigeria and serve clients across West Africa and the broader African continent. Our services are designed to meet NAFDAC, WHO, EMA, ICH, and FDA requirements.
Our clients include pharmaceutical companies, marketing authorisation holders (MAHs), contract research organisations (CROs), healthcare institutions, and regulatory stakeholders across Nigeria and West Africa.
Yes, IntraHub Africa is registered with NAFDAC and maintains up-to-date professional body memberships and regulatory registrations. Please contact us for full details of our current accreditations and certifications.
Simply book a free consultation. A member of our team will schedule a no-obligation call to understand your products, markets, and current pharmacovigilance pain points. From there, we conduct a needs assessment and present a tailored roadmap.